i-STAT Troponin I/cTnI Cartridge
Regular price
$629.95
$499.00 USD
21% OFF
ONLY LEFT
Abbott Cartridge PROVIDING LAB-QUALITY RESULTS IN MINUTES
A wide range of cartridges for diagnostic testing at the bedside
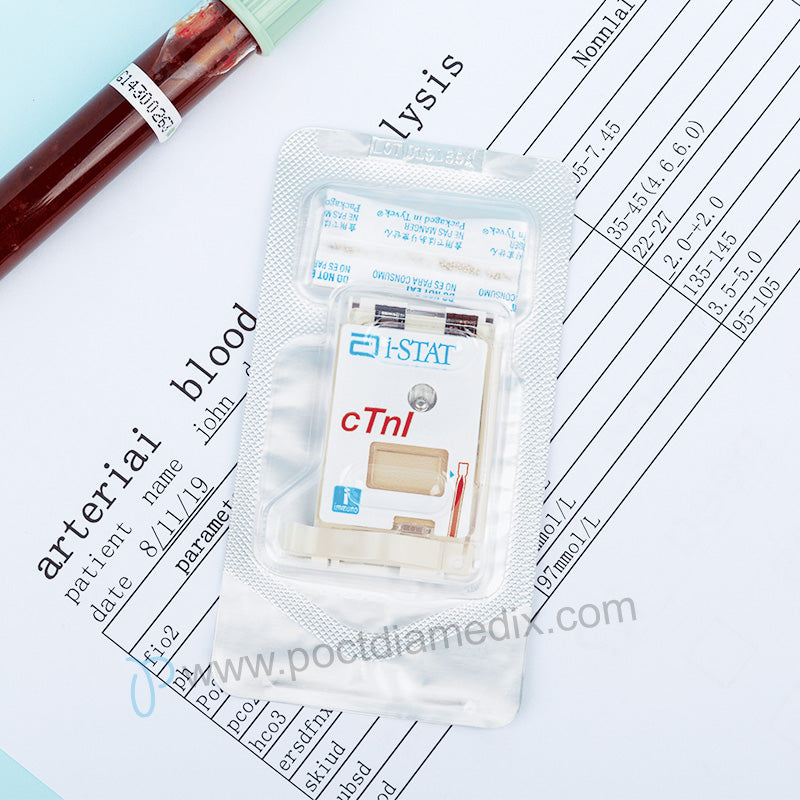
- The i-STAT cardiac troponin I (cTnI) test is an in vitro diagnostic test for the quantitative measurement of cardiac troponin I (cTnI) in whole blood or plasma. Measurements of cardiac troponin I are used in the diagnosis and treatment of myocardial infarction and as an aid in the risk stratification of patients with acute coronary syndromes with respect to their relative risk of mortality.
-
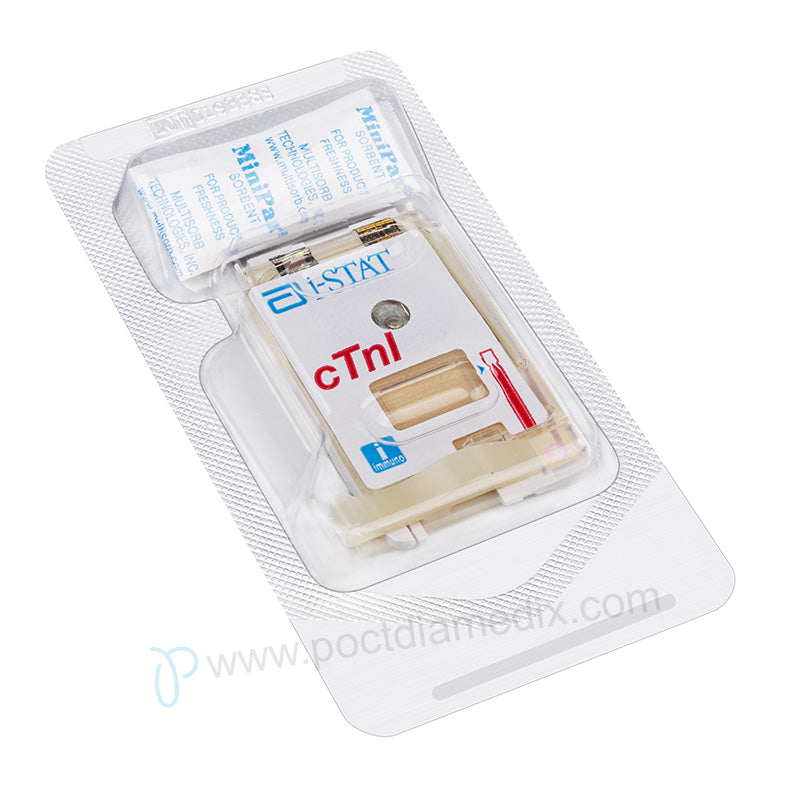
i-STAT Cartridge Test Troponin. NOTE: This product is not CLIA-waived; it is CLIA-classified as moderately complex.
FDA-cleared for professional use only. - Sample Type: cTnI cartridges require the use of heparinized whole blood or plasma, or nonheparinized whole blood tested within one minute of patient draw.
- Sample Volume:17 μL
- Analysis Time:cTnI cartridges: 600 seconds (10 min.)
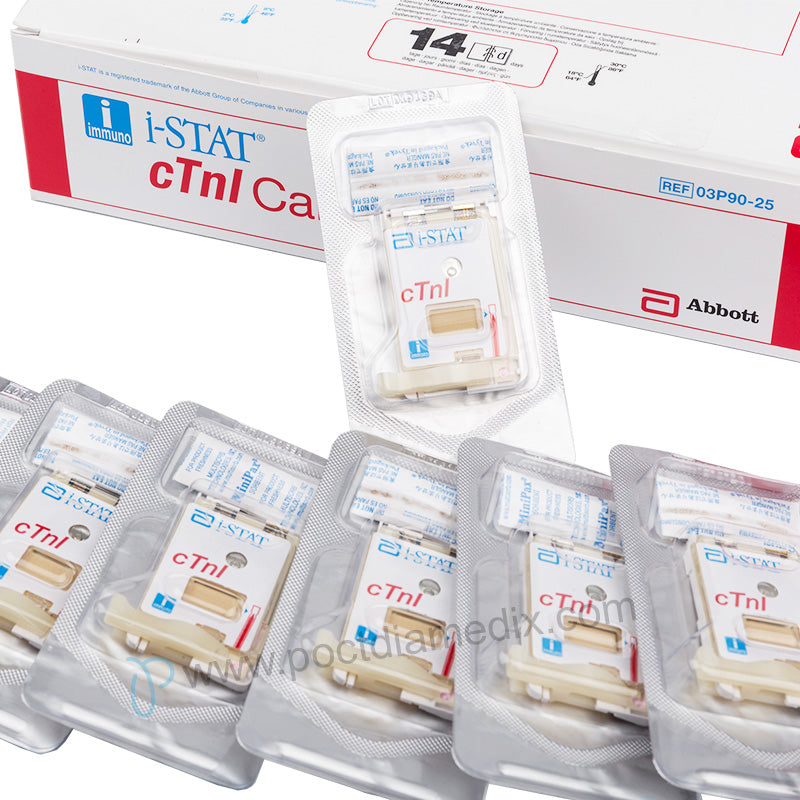
- Package:25 Tests
- Shelf Life:Refrigerated at 2 to 8ºC (35 to 46ºF) until expiration date.
- Room temperature at 18 to 30ºC (64 to 86ºF) for two weeks.
- NO RETURNS ;REFUNDS or EXCHANGES ARE ACCEPTED AFTER ORDER IS PLACED BY ONLINE.
-
Note Regarding System Reliability:The i-STAT 1 System automatically runs a comprehensive set of quality checks of analyzerand cartridge performance each time a sample is tested. This internal quality system willsuppress results if the analyzer or cartridge does not meet certain internal specifications(see Quality Control section in System Manual for detailed information). To minimize theprobability of delivering a result with medically significant error the internal specificationsare very stringent. It is typical for the system to suppress a very small percentage ofresults in normal operation given the stringency of these specifications. If however theanalyzer or cartridges have been compromised, results may be persistently suppressed,and one or the other must be replaced to restore normal operating conditions.
-
The cTnI cartridges can only be used with the i-STAT 1 analyzer bearing the symbol.
-
*For in vitro diagnostic use only.
people are currently looking at this product
Estimated delivery time 3-5 days
Guaranteed Safe Checkout